Compressed air purity is critical in medical manufacturing, pharmaceutical production, laboratory environments, and clean-processing facilities. Any contaminants in compressed air can affect product sterility, equipment performance, and patient safety. ISO 8573 is the internationally recognised standard that defines air quality limits for particle concentration, moisture, and oil content, providing a consistent framework for compliance and system design across medical and pharmaceutical applications in Australia.
Breaking Down ISO 8573 Air Quality Classes
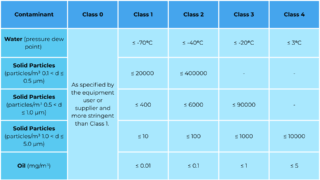
ISO 8573 classifies compressed air purity across three key contaminant categories: solid particles, water, and total oil (aerosol + vapour + liquid). The standard allows facilities to specify the required cleanliness level for their processes.
Medical, biotech, and pharmaceutical environments typically require very low oil content due to contamination risk. Many Australian facilities operate under:
- ISO 8573 Class 0 or Class 1 for oil
- Tightly controlled humidity levels for moisture
- Very low solid particle thresholds for sterile manufacturing

Choosing the correct ISO class ensures compliance with internal quality systems, regulatory inspections, and global manufacturing standards.
Designing Systems to Meet ISO 8573
Meeting ISO 8573 requirements depends on correct compressed air system design. Key elements include:
- Oil-free compressor selection to eliminate upstream oil introduction
- Primary and secondary filtration stages targeting coarse and fine particulate removal
- Refrigerant or desiccant drying systems to achieve required moisture levels
- Stainless-steel or clean-air-rated piping to avoid internal corrosion or oil-carryover
- Continuous or periodic air quality monitoring supporting traceability and quality control

System design must also consider external contamination sources such as lubricant migration, airborne dust at intake, and bacterial presence in humid environments.
Compliance, Verification, and Ongoing Testing
Maintaining ISO 8573 compliance is not a one-time activity. Facilities should incorporate:
- Routine ISO 8573 air sampling and analysis
- Scheduled testing intervals aligned with QA requirements
- Documentation for regulatory audit compliance
- Preventative maintenance and filter replacement cycles
Independent verification supports Good Manufacturing Practice (GMP) standards, Therapeutic Goods Administration (TGA) and Food and Drug Administration (FDA) compliance considerations, ISO 9001 quality frameworks, and cleanroom operational requirements.
Pneumatic Engineering (PE) can assist with compliant system design, ISO 8573 testing, ongoing maintenance, and ensuring that your compressed-air infrastructure meets required medical and pharmaceutical quality levels.
ISO 8573 provides a standardised approach to compressed-air quality that is essential for sterile production, patient safety, and product reliability. By selecting the correct system architecture, applying appropriate filtration and drying technologies, and performing regular compliance testing, medical and pharmaceutical companies can confidently meet their regulatory and operational requirements. Pneumatic Engineering is ready to support your facility with compliant compressed-air systems, quality testing, upgrades, and technical advice.




